Carthamin Red Dye
November 29, 2021
Everyone has heard the
mantra, "
Dress for Success." This
idea has been around for
millennia. In the
ancient world, the
higher classes wore
clothing dyed with
Tyrian purple, also known as royal purple, and now known
chemically as
6,6'-dibromoindigo (C16H8Br2N2O2). This dye was
harvested as a
secretion produced by
sea snails in the
family Muricidae. It was
expensive, since many snails were required and the extraction involved much
labor. So close was the relationship between this dye color and status, that eventually only the
Roman emperor was permitted to wear Tyrian purple.
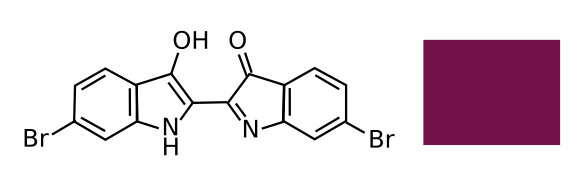
Chemical structure of (C16H8Br2N2O2), known as Tyrian purple. The IUPAC name of this compound, usually called 6,6'-dibromoindigo, is an impressive 6-bromo-2-(6-bromo-3-hydroxy-1H-indol-2-yl)indol-3-one. The hexadecimal color code, as shown in the color patch, is 66023C. (Created using Inkscape. Click for larger image.)
The use of Tyrian purple is traced as early as 1,570
BC in
Phoenicia. The
Greek historian,
Theopompus (Θεόπομπος, c.380 BC - c.315 BC) reported that Tyrian purple was worth its
weight in
silver.
Aristotle (384 BC - 322 BC) described in his
History of Animals the source of Tyrian purple and its process of extraction.
Pliny the Elder (23 AD - 79 AD) described the production of Tyrian purple in Book IX of his
Natural History. About 2,500 sea snails were need to
produce just an
ounce of the dye.
Before the advent of
chemical synthesis,
Nature was our only source of
colorants. The following table lists some of the many natural sources of colorants.
Some of the many plants that can be used for dyes[1-2]
| Dye Color |
Plant Common Name (Additional Colors) |
| Yellow Dyes |
Honey Locust |
| Indiangrass (brown, green) |
| Safflower (red) |
| Gardenia (red, blue) |
| Turmeric |
| Orange Dyes |
Bloodroot (brown, yellow) |
| Sassafras (black, green, purple, yellow) |
| Red Dyes |
Sumac (yellow, green, brown, black) |
| Chokecherry |
| Slippery Elm (brown, green, yellow) |
| Beetroot |
| Grape (blue-violet) |
| Purple / Blue Dyes |
Red Mulberry (brown, yellow, green) |
| Summer Grape (orange, yellow, black) |
| Black Locust (black, green, yellow, brown) |
| Green Dyes |
Stinging nettle |
| Goldenrod (yellow, brown) |
| Chlorophyll |
| Gray Dyes |
Butternut (brown) |
| Brown Dyes |
Elderberry (yellow) |
| Cocoa |
| Tamarind |
| Black Dyes |
Sumac (yellow, red, green, brown) |
The first
synthetic dye,
mauveine, was
discovered accidentally by a very
young William Henry Perkin (1838-1907) in 1856. Since it was an
aniline derivative, it's also known as aniline purple. Interestingly, the
chemical structure of mauveine was unknown until 1994.[3] Perkin made the discovery while assisting
chemist,
August Wilhelm von Hofmann (1818-1892), in
research on synthetic
quinine, but he made the discovery in his
home laboratory independently of Hofmann. This is reminiscent of the home laboratory discovery of
xerography by
Chester Carlson (1906-1968).

William Henry Perkin (1838-1907) in 1906 is shown on the left, with a photograph of a patch of mauveine dyed silk that he made at the center, and a box on the right that's colored with the acknowledged #8D029B hexadecimal color code for mauveine. (Left image, a 1906 photo of William Henry Perkin from A History of Chemistry (1918) by F. J. Moore. Center image, a portion of a photograph by Henry Rzepa. The left and center images are from Wikimedia Commons. Click for larger image.)
The
safflower,
Carthamus tinctorius, produces a range of colorants from yellow to red, and it's
cultivated mostly for its
seeds that are used to make
vegetable oil. Dried safflower
flowers are a source for the orange-red dye,
carthamin, also known as Natural Red 26. Safflower
petals are yellow to orange in color until full bloom, and they change to a reddish color upon
wilting.
Carthamin has been used
worldwide for more than 4500
years and it's the major component of the
Japanese textile dye called
beni. In a recent study, a team of Japanese
scientists from
Tohoku University (Sendai, Miyagi, Japan),
Toyo Ink SC Holdings Co. (Saitama, Japan),
TOYOCHEM Co., Ltd (Tokyo, Japan), and the
Tohoku Medical Megabank Organization (Sendai, Miyagi, Japan) has identified the
genes coding for the
enzyme responsible for the formation of carthamin from a
compound known as precarthamin, a
water-
soluble quinochalcone.[4-5] An explanation was also developed that explains how the red
pigmentation of safflower petals forms during flower
senescence.[4]
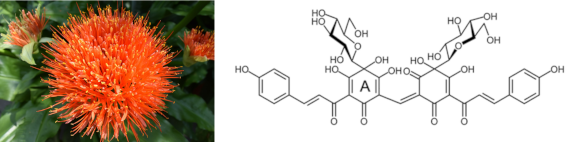
(Red-orange safflower and the chemical structure of the red dye, carthamin. Left image via Wikimedia Commons. Right image, ©Toshiyuki Waki et al. Click for larger image.)
Although carthamin dye has been used for more that 4,000 years, it wasn't until the late
19th and early
20th centuries that scientists started to examine its chemical structure.[5] The present study identified the genes for an enzyme that's responsible for the final step of carthamin
biosynthesis called "carthamin
synthase."[5] Carthamin synthase is similar to
peroxidase, an enzyme found in such plants as
turnips and
radishes. It uses
molecular oxygen instead of the
hydrogen peroxide of peroxidase as a
hydrogen acceptor in its formation of carthamin.[5] Carthamin synthase transforms the safflower yellow to red.[5]
Says study co-author,
Toru Nakayama, of Tohoku University,
"In the future, we may be able to produce this useful compound [carthamin] microbially in large amounts using metabolic engineering."[5]
References:
- Native Plant Dyes, U.S. Forest Service of the United States Department of Agriculture.
- List of colorants derived from natural products, TOYOCHEM CO., LTD.
- Otto Meth-Cohn and Mandy Smith, "What did W. H. Perkin actually make when he oxidised aniline to obtain mauveine?" Journal of the Chemical Society, Perkin Transactions, vol. 1 (1994), pp. 5-7, doi:10.1039/P19940000005.
- Toshiyuki Waki, Miho Terashita, Naoki Fujita, Keishi Fukuda, Mikiya Kato, Takashi Negishi, Hiromi Uchida, Yuichi Aoki, Seiji Takahashi, and Toru Nakayama, "Identification of the Genes Coding for Carthamin Synthase, Peroxidase Homologs that Catalyze the Final Enzymatic Step of Red Pigmentation in Safflower (Carthamus tinctorius L.)," Plant and Cell Physiology, vol. 122 (August 26, 2021), https://doi.org/10.1093/pcp/pcab122.
- Researchers Identify the Biosynthesis of Carthamin, the Historic Red Colorant in Safflowers, Tohoku University Research News. September 2, 2021.
Linked Keywords: Mantra; Dress for Success; idea; millennium; millennia; ancient history; ancient world; nobility; higher classes; clothing; dye; Tyrian purple; chemistry; chemically; 6,6'-dibromoindigo (C16H8Br2N2O2); harvest; harvested; secretion; sea snail; family (biology); Muricidae; cost; expensive; employment; labor; Roman emperor; International Union of Pure and Applied Chemistry; IUPAC; chemical compound; hexadecimal; web color; hex triplet; color code; Inkscape; Anno Domini; BC; Phoenicia; Ancient Greece; Greek; historian; Theopompus (Θεόπομπος, c.380 BC - c.315 BC); weight; silver; Aristotle (384 BC - 322 BC); History of Animals; Pliny the Elder (23 AD - 79 AD); Natural History (Pliny); production (economics); ounce; chemical synthesis; Natural dye; colorant; Honey Locust; Sorghastrum nutans; Indiangrass; Safflower; Gardenia; Turmeric; Sanguinaria; Bloodroot; Sassafras; Sumac; Prunus virginiana; Chokecherry; Ulmus rubra; Slippery Elm; Beetroot; Grape; Morus rubra; Red Mulberry; Vitis aestivalis; Summer Grape; Robinia pseudoacacia; Black Locust; Urtica dioica; Stinging nettle; Goldenrod; Chlorophyll; Juglans cinerea; Butternut; Sambucus; Elderberry; Cocoa; Tamarind; synthetic; mauveine; serendipity; discovered accidentally; youth; young; William Henry Perkin (1838-1907); aniline; derivative (chemistry); structural formula; chemical structure; chemist; August Wilhelm von Hofmann (1818-1892); research; quinine; home; laboratory; xerography; Chester Carlson (1906-1968); digital photography; photograph; silk; Henry Rzepa; Wikimedia Commons; Carthamus tinctorius; horticulture; cultivate; seed; vegetable oil; flower; carthamin; petal; wilting; worldwide; year; Japan; Japanese; textile; scientist; Tohoku University (Sendai, Miyagi, Japan); Toyo Ink SC Holdings Co. (Saitama, Japan); TOYOCHEM Co., Ltd (Tokyo, Japan); Tohoku Medical Megabank Organization (Sendai, Miyagi, Japan); gene; molecular genetics; coding; enzyme; chemical compound; aqueous solution; water; solubility; soluble; chalcone; quinochalcone; pigment; pigmentation; cellular senescence; chemical structure of the red dye, carthamin; 19th century; 20th century; biosynthesis; synthase; peroxidase; turnip; radish; molecule; molecular; oxygen; hydrogen peroxide; hydrogen; electron acceptor; Toru Nakayama; microorganism; microbial; metabolic engineering.