Seeing Infrared
January 19, 2015
Infrared light has been
technologically useful for many things. Short-range
data communication by infrared light in the 850–900
nanometer range is used in many devices, including the ubiquitous
remote control. As in many
consumer technologies, advances in functionality were obtained though
standardization; in this case, the
Infrared Data Association (IrDA) standard.

A collection of infrared remote control devices in my house. The Panasonic remote for our legacy videocassette recorder is seldom used.
(Photo by author)
A typical
human eye will respond to
wavelengths from about 390 to 720 nm, so we can't see into the infrared. The
lens of the eye blocks
ultraviolet light, and when the
lens is absent, vision to wavelengths as low as 300 nm has been reported.
Although infrared light is useful for many applications, it's often converted to
visible light by
optical frequency doubling using the
optical non-linearity of some materials. Doubling of
frequency is equivalent to reduction of the wavelength by half. I wrote about frequency doubling in two previous articles (Second Harmonic Light Generation, September 30, 2011 and
Thin Optical Doubler, July 23, 2014).
Efficient optical doubling can be achieved in
crystals of lithium niobate (LiNbO3), lithium tantalate (LiTaO3), potassium titanyl phosphate (KTP, KTiOPO4), lithium triborate (LBO, LiB3O5) and β-barium borate (BBO, β-BaB2O4). These non-linear optical effects only occur at high light intensities, where the
electric field component of the light is correspondingly high, so it only works for laser light sources.
Efficient conversion occurs only for a long
optical path through the doubling crystal. As an example, the
solid state laser, Neodymium-YAG (Nd:YAG), emits light at the infrared wavelength of 1.064 μm. A nonlinear optical crystal will double this 1.064 μm light to 532 nm, which is green light.
Lasers and frequency-doublers aren't the only way to convert infrared to visible light. Another method is the use of an upconversion phosphor. The simplest such process is
excited state absorption in which an
electron, already excited to a higher
energy level by a
photon, is excited to a still higher level by a second photon. When the electron again seeks its
ground state, it emits visible light.
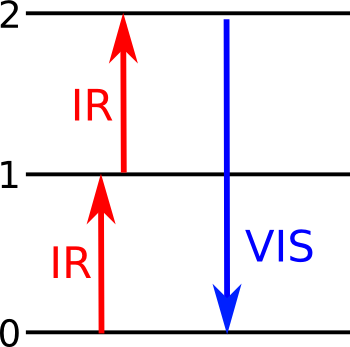
Energy level diagram for upconversion via excited state absorption.
An electron at a ground state (0) is excited to a first (1) energy level by an infrared photon, and a higher, second (2) energy level by another photon. When it reverts back to its ground state, visible light is emitted.
(Illustration by the author, rendered with Inkscape.)
A more useful form of upconversion is
energy transfer upconversion in which infrared photons are absorbed by two different
atom types, an "activator" ion and a "sensitizer" ion. As shown in the figure,
non-radiative transfer of energy from the sensitizer atom to the activator electron allows it to achieve a higher energy state than it could have through absorption of a single infrared photon. One useful activator-sensitizer combination is
Er3+ and
Yb3+.[1-2]
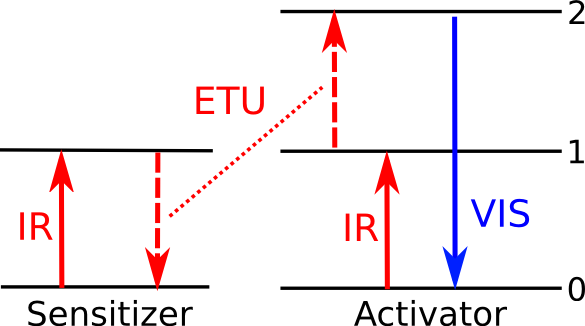
Energy level diagram for upconversion via energy transfer. An electron at a ground state (0) in an activator ion is excited to a first (1) energy level by infrared light, and a second (2) energy level with help from non-radiative energy transfer from a sensitizer ion. When it reverts back to its ground state, visible light is emitted. (Illustration by the author, rendered with Inkscape.)
Since ultraviolet light is useful as an
antimicrobial agent, there has also been
research done on upconversion of visible light to ultraviolet light.[3] Since an upconversion phosphor converts two infrared photons to a single visible photon, it can have an apparent efficiency greater than one. Upconversion phosphors will saturate when their excited state is filled because of its long
lifetime.
The human eye is a sensitive
photodetector, but it's always been thought that it was not possible for the eye to detect infrared. A quick glance at the response of the
retinal cones to light of various wavelengths (see figure) shows that there is no vision above about 720 nanometers. However, a large international team of
scientists from
Polgenix, Inc. (Cleveland, Ohio), the
Washington University School of Medicine (St. Louis, Missouri), the
Nicolaus Copernicus University (Torun, Poland), the
University of Bern (Bern, Switzerland),
Case Western Reserve University (Cleveland, Ohio), and the
University of Oslo (Oslo, Norway), has just
published evidence of human vision at wavelengths beyond 1,000 nm.[4-5]
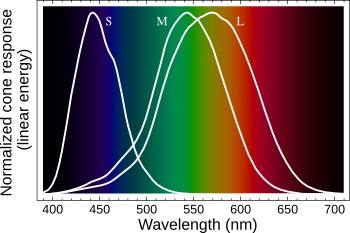
Visual response of the retinal S, M and L cone cells that define color vision.
(Modified Wikimedia Commons image.)
There have been
anecdotal reports of scientists seeing flashes of green light while working with infrared lasers, an effect that would indicate frequency doubling but had not been explained.[4-5] Says
Frans Vinberg, a
postdoc in the
Department of Ophthalmology and Visual Sciences at Washington University and lead
author of the study,
"They were able to see the laser light, which was outside of the normal visible range, and we really wanted to figure out how they were able to sense light that was supposed to be invisible."[5]
Vinberg and his
colleagues combed the
scientific literature to see what conditions resulted in human observation of infrared, and they conducted
psychophysical experiments that proved the effect.[4-5] In their experiments, the research team beamed
pulses of infrared light into the eyes of their subjects. By varying the pulse width, but keeping the total number of photons in each pulse constant, they found that a rapid influx of photons resulted in two infrared photons teaming to produce a green light photon.[5]
The sensitivity increased at wavelengths above 900 nm. Doubled 900 nm light would be at 450 nm, which is at the lower range of optical sensitivity. The effect displayed a
quadratic dependence on laser power, an indicator of a nonlinear optical process.[4] Other experiments showed that infrared light causes a
photoisomerization of a model
chromophore compound, which indicates that human perception of infrared light occurs by such an isomerization of visual
pigments.[4]
This strange visual
phenomenon might have
diagnostic value for
diseases such as
macular degeneration, since it would allow
physicians to stimulate the retina in a new way.[5] This research was funded by the
National Institutes of Health (NIH) and other agencies.[5]
References:
- N. Menyuk, K. Dwight and J.W. Pierce, "NaYF4 : Yb,Er—an efficient upconversion phosphor," Appl. Phys. Lett., vol. 21, no. 4 (August 15, 1972), pp. 159 ff. http://dx.doi.org/10.1063/1.1654325
- Yanmin Yang, Chao Mi, Fuyun Jiao, Xianyuan Su, Xiaodong Li, Linlin Liu, Jiao Zhang, Fang Yu, Yanzhou Liu, and Yaohua Mai, "A Novel Multifunctional Upconversion Phosphor: Yb3+/Er3+ Codoped La2S3," Journal of the American Ceramic Society, vol. 97, no. 6 (June, 2014), pp. 1769–1775.
- Ezra Lucas Hoyt Cates, Dissertation: Development of Visible-to-Ultraviolet Upconversion Phosphors for Light-Activated Antimicrobial Technology, Georgia Institute of Technology, May, 2013.
- Grazyna Palczewska, Frans Vinberg, Patrycjusz Stremplewski, Martin P. Bircher, David Salom, Katarzyna Komar, Jianye Zhang, Michele Cascella, Maciej Wojtkowski, Vladimir J. Kefalov, and Krzysztof Palczewski, "Human infrared vision is triggered by two-photon chromophore isomerization," Proc. Natl. Acad. Sci., (Online Early Edition, December 1, 2014), doi:10.1073/pnas.1410162111.
- Jim Dryden, "The human eye can see 'invisible' infrared light," Washington University School of Medicine Press Release, December 1, 2014.
Permanent Link to this article
Linked Keywords: Infrared light; technology; technological; data; communication; nanometer; remote control; consumer electronics; consumer technologies; standardization; Infrared Data Association (IrDA); remote control device; Panasonic; videocassette recorder; human eye; wavelength; lens of the eye; ultraviolet light; aphakia; visible light; second-harmonic generation; optical frequency doubling; nonlinear optics; optical non-linearity; material; frequency; wavelength; energy conversion efficiency; crystal; lithium niobate; lithium tantalate; potassium titanyl phosphate; lithium triborate; β-barium borate; intensity; electric field; Poynting vector; laser; optical path; solid state laser; Nd:YAG; Neodymium-YAG; micrometer; upconversion; phosphor; excited state; electron; energy level; photon; ground state; energy level diagram; Inkscape; energy transfer upconversion; atom; non-radiative transfer of energy; erbium; Er3+; ytterbium; Yb3+; antimicrobial agent; research; lifetime; photodetector; cone cells in the human eye; retinal cones; scientist; Polgenix, Inc. (Cleveland, Ohio); Washington University School of Medicine (St. Louis, Missouri); Nicolaus Copernicus University (Torun, Poland); University of Bern (Bern, Switzerland); Case Western Reserve University (Cleveland, Ohio); University of Oslo (Oslo, Norway); academic publishing; retina; retinal; S, M and L cone cells; Wikimedia Commons; anecdotal evidence; Frans Vinberg; postdoctoral research; postdoc; Department of Ophthalmology and Visual Sciences; author; colleague; scientific literature; psychophysics; psychophysical; experiment; pulse; quadratic function; photoisomerization; chromophore compound; pigment; phenomenon; diagnosis; diagnostic; disease; macular degeneration; physician; National Institutes of Health.