Entropic Order
August 20, 2012
You've likely performed this process for yourself, many times. You have some objects in a pile in the middle of a sheet; for example,
frozen French fries on a baking sheet. You shake the sheet to organize the pile into a
two-dimensional array; and, with a little extra shaking, you can arrange most of the French fries to be
parallel to each other.
There aren't any
chemical or
physical attractive forces that allow this process. The shape of the French fries, alone, suffices. This is an example of
entropic order. Your system moves into a lower
energy state that's easier to fall into than to get out of.

Do you want entropy with that?
Physics is truly in everything around us, even French fries.
(Via Wikimedia Commons))
An essential step in this process is the shaking. Adding
noise to a system to achieve a goal is used in several other processes; for example,
metallurgical annealing, it's computational cousin,
simulated annealing, and
stochastic resonance. Metallurgical annealing is the process wherein a
material is subjected to a
temperature just below its
melting point. This allows its constituent
atoms to move around and establish a different
equilibrium state with different
mechanical properties.
Simulated annealing is the
computational analog of physical annealing in which the interaction between particular data objects is a function of a
variable that's analogous to temperature. The "attractive forces" between the data objects are small at high temperature, so the attraction is small, and the data are jumbled. As the temperature is decreased, the data objects start to attach themselves to each other. About twenty-five years ago, I wrote a
sorting program based on simulated annealing. The program did accomplish sorting, but it made
bubble sort seem speedy.
Another place where shaking helps is when the shaking is
electrical noise. In 1982 it was realized that adding noise sometimes aids in the detection of
periodic signals.[1] The observation was framed in the context of how small changes in the
orbit of the Earth could cause the
ice ages. The idea was that the Earth's
climate had two stable states, one at low temperature and another at high temperature. A small additional forcing would cause a flip from one state to another.[2]
This theory of the ice ages was not supported by other data, but this idea was shown to be valid for
bistable electrical circuits and
ring laser resonators. The idea that adding noise to a system can enable transitions between system states is known as stochastic resonance. It should be noted that you need just the right amount of noise. Too little, or too much noise won't work. The figure below shows how a digital signal below a threshold can be recovered by adding noise.
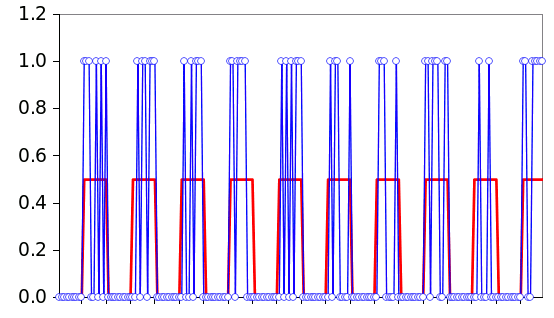
Gnumeric. The source file is available here)
Researchers from the
Applied Physics Program, the
Department of Chemical Engineering, and the
Department of Materials Science and Engineering of the
University of Michigan (Ann Arbor, Michigan) have investigated the
crystallization of shaped
nanoparticles and reported on their research in a recent issue of
Science.[3-4] This study was a collaborative effort between the various specialties of the study's three authors.
Sharon Glotzer is a
physicist and
chemical engineering professor,
Michael Engel is a
chemical engineer who wrote the
computer code, and applied physics graduate student
Pablo Damasceno ran the thousands of virtual
experiments required for the study.[3]
The University of Michigan team did
computer simulations of ordering for 145 types of
convex polyhedra when numbers of each type were crowded into a small
volume.[4]. Nearly seventy percent of these polyhedral shapes produced crystalline structures, and the only driving force was entropy, since there were no attractive forces built into the model.[3] The crystal structures ranged from the simple structure found in
rock salt, to complex structures with 52 particles in a
unit cell, the smallest repeatable volume of a
crystal.[3] Some of the structures were "plastic" crystals for which particles could spin in place.[3]

Shaped particles forming a crystal, an example of entropic ordering from the University of Michigan study.
(University of Michigan image/P. Damasceno, M. Engel, S. Glotzer))
Glotzer explained the entropic driving force with reference to a
thought experiment in which you fill a jar with
dice in a
gravity-free environment. With just a few dice in the jar, the floating dice would be oriented
randomly. As you add dice, the accessible volume becomes so small that the dice have a more limited range of orientation options. Finally, the dice are forced to align face-to-face.[3]
The research team notes that the shape of the individual nanoparticles predicts the resultant crystal structure.[3] This leads to an interesting route for
mechanical alloying when the particles are of the same shape, but formed of different materials. Just as interesting are the thirty percent of polyhedra that didn't form a crystal. The University of Michigan research was funded by the
U.S. Department of Defense, the
US Department of Energy, the
National Science Foundation, and
Deutsche Forschungsgemeinschaft.[3]
Another of my articles (Depletion Attraction, March 7, 2011) was about a similar
thermodynamic ordering mechanism that doesn't require the particles themselves to provide force. In the case of depletion attraction, the particles are of different sizes, and the driving force arises from the fact that large particles have a large volume but a small ratio of surface area to volume.
Many small particles filling the same volume as one larger particle will have a larger surface area exposed to the solution in which they reside. Larger particles stick to each other, simply because the small particles that push them around in
solution can't access the space between closely spaced large particles. Instead, the smaller particles have nothing else to do than to push the larger particles towards each other.
References:
- Roberto Benzi, Alfonso Sutera and Angelo Vulpiani, "The mechanism of stochastic resonance," J. Phys. A: Math. Gen., vol. 14 (1981), pp. L453-L457.(PDF File); R. Benzi, G. Parisi, A. Sutera, A. Vulpiani, "Simple climate models with periodic and stochastic forcing," Tellus, vol. 34 (1982), p.10.
- L. Gammaitoni , P. Hänggi , P. Jung and F. Marchesoni, "Stochastic Resonance: A remarkable idea that changed our perception of noise," Eur. Phys. J., vol. B 69 (2009), pp. 1–3 (PDF File).
- Nicole Casal Moore, "Entropy can lead to order, paving the route to nanostructures," University of Michigan Press Release, Jul 26, 2012.
- Pablo F. Damasceno, Michael Engel and Sharon C. Glotzer, "Predictive Self-Assembly of Polyhedra into Complex Structures," Science, vol. 27, no. 6093 (July 27, 2012), pp. 453-457.
Permanent Link to this article
Linked Keywords: Frozen food; French fries; two-dimensional space; two-dimensional; array; parallel; chemical bond; Coulomb's law; physical attractive force; entropy; entropic; energy level; energy state; physics; Wikimedia Commons; randomness; noise; metallurgical annealing; simulated annealing; stochastic resonance; material; temperature; melting point; atoms; thermodynamic equilibrium; equilibrium state; mechanics; computation; variable; sorting algorithm; sorting program; bubble sort; electrical noise; periodic signal; orbit of the Earth; ice age; climate; flip-flop; bistable electrical circuit; ring laser resonator; Gnumeric; Applied Physics Program; Department of Chemical Engineering; Department of Materials Science and Engineering; University of Michigan (Ann Arbor, Michigan); crystallization; nanoparticle; Science; Sharon Glotzer; physicist; chemical engineering; professor; Michael Engel; chemical engineer; source code; computer code; Pablo Damasceno; experiment; computer simulation; convex polyhedra; volume; rock salt; unit cell; crystal structure; crystal; thought experiment; dice; weightlessness; gravity-free environment; randomness; random; mechanical alloying; U.S. Department of Defense; US Department of Energy; National Science Foundation; Deutsche Forschungsgemeinschaft; depletion attraction; thermodynamics; solution.