Energy from Evaporation
July 16, 2015
My first lesson in the
thermodynamics of
evaporation came from my
high school chemistry teacher. As most people know, teacher's
salaries aren't all that high. This teacher lived with his
family in the
rented upstairs
apartment of a
two-family house, since he couldn't afford his own
house. His
heating was supplied as part of his rent, but his
thermostat temperature adjustment was fixed at a setting too low for his preference.
His
trick for getting more heat when he felt too cold was to place an
alcohol-soaked rag over the thermostat. The evaporation of the alcohol caused a local cooling that tricked the device into giving more heat. The
physical mechanism for this is the
latent heat of
vaporization (also called the
enthalpy of vaporization) combined with the temperature change associated with
heat capacity.

The Honeywell "round" thermostat.
This device, patented as US Patent No. 2,394,920 on February 12, 1946, was sold for many years with a sealed mercury switch activated by a temperature-sensitive bimetallic strip.
(Via Wikimedia Commons.)
The latent heat of vaporization of
isopropyl alcohol (IPA) at "
standard conditions" of 298.15
K and 1
atm is about 45
kilojoule per mole (kJ/mol).[1] The "
rubbing alcohol" used by my chemistry teacher is not 100% isopropyl alcohol; rather, it's a
mixture of
water with only about 70% alcohol. In that case, one
milliliter contains about 0.009
moles of IPA.
The heat capacity of water is about 4.18 J/g-K, and the heat capacity of IPA is about 2.68 J/g-K. As you can see, this is turning into a major
calculation, since the
concentration of IPA and the
volume of
solution will change over time. It's time for a rough
estimate. Cranking through some numbers reveals a cooling of at least 10
°C when a milliliter of the mixture is evaporated. The evaporation happens slowly, so we can expect our teacher's thermostat can be fooled into thinking a room is a few
degrees Fahrenheit cooler than it really is.
This simple evaporation process could be used as a costly and
inefficient energy source by spreading the alcohol on a
thermopile to convert a temperature difference to an
electric current. However, if you're trapped in a
bar during a
hurricane and happen to have a
thermoelectric generator in your pocket, you might power a
radio with a bottle of
vodka.
Such a thing could be done less expensively, but less efficiently, with water. Water has an
enthalpy of vaporization of about 44 kJ/mol, nearly identical to that of IPA. However, the
vapor pressure of water at
room temperature is just 2.3 kPa, compared with IPA's 6 kPa, so its evaporation rate is slower.

The drinking bird toy is powered by the evaporation of water.
Evaporation of water from the felt-covered beak causes dichloromethane to condense because of the lowered temperature.
(Via Wikimedia Commons.)
A team of
scientists and
engineers from
Columbia University (New York, New York), with an assist from a
microbiologist at
Loyola University (Chicago, Illinois), has taken an alternative route to extraction of
mechanical energy from the evaporation of water. They use the property of
bacterial spores that they shrink and swell with changing
humidity. In that way, they can be used to exert a mechanical
force to push and pull other objects.[2-]
As anyone can see while looking outside after a warm
summer's rainfall, evaporation is an ubiquitous
natural phenomenon. It's a major form of
energy transfer affecting
Earth's climate. Says
Ozgur Sahin, an
associate professor of
biological sciences and
physics at Columbia University and the study's lead
author,
"Evaporation is a fundamental force of nature... It's everywhere, and it's more powerful than other forces like wind and waves... Our climate is powered by evaporating water from the oceans, and we have no direct way of accessing this energy."[3,6]
There's been previous
research on
materials that respond mechanically to
chemical stimuli, and there have also been
biomimetic systems that
oscillate, transport
fluid, and change shape. These materials have been far less efficient at generating
work compared with conventional
actuators.[5] Last year, researchers from Columbia University,
Harvard University, and Loyola University teamed in a demonstration that
Bacillus spores exert a mechanical response of 10 MJ/m
3 to water
gradients.[5] This is a thousand times more force than
human muscles and a hundred times greater than
synthetic water-responsive materials.[5-6]
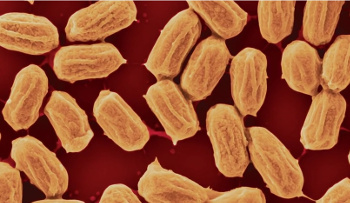
Bacillus bacterial spores.
Bacillus bacteria are a common soil microorganism.
(Still image from a YouTube Video.)[)
As a
survival mechanism, the Bacillus microorganism forms a rigid, dormant spore when
starved. When these spores are exposed to humidity, they expand up to 40% in volume, and the effect is
reversible.[6] As Sahin explains,
"Changing size this much is highly unusual for a material that is as rigid as wood or plastic... We figured that expanding and contracting spores can act like a muscle, pushing and pulling other objects. We noticed that we could harness the motion of spores and convert it to electrical energy."[6]
In last year's study, the research team identified
mutations that almost double the energy density of the spores. They were also able to get the spores to
self-assemble into dense,
submicrometer-thick monolayers on various
substrates, including sheets of
elastomer and
silicon microcantilevers.[5] In that first study, the research team demonstrated the utility of their material by building an
energy-harvesting device in which movement of a
flexing membrane creates
electricity by moving a
magnet inside a
coil of wire (see figure).[5]

Even scientists play with Lego®!
In this device, a spore-coated rubber sheet moves a magnet inside a coil of wire to produce electricity.
(Columbia University image by Xi Chen.)
In the recently
published study, the researchers have produced additional devices in which the spores are used for
locomotion and electricity generation. The devices, which generate
piston and
rotary motion, start spontaneously when placed in the humid region above a water surface.[2-3] There's a piston coupled to an electrical generator that produces enough
power to flash a
light-emitting diode,[3] and a 100
gram miniature car powered by the evaporation of a contained water source (see figure).[2]

The evaporation-powered car, called "Eva," is propelled by a turbine engine fueled by water evaporating from wet paper.
(Columbia University image by Xi Chen.)
While the car's engine concept, called a "moisture mill," could be used as an electrical generator, a much better
device architecture is found in their version of an
artificial muscle. They constructed such muscles by
printing spores in dashed patterns on both sides of a plastic tape. Dry air shrinks the spores, and the tape converts from straight to wavy, while humidity reverses the process to straighten the tape. Many of these tapes are
paralleled to produce a greater force, and movement of the tapes also controls
shutters to modulate the humidity and cause a piston oscillation.[3]

The Evaporation Engine floats on water, and its spore mechanism creates a piston movement capable of generating electricity.
(Columbia University image by Xi Chen.)
Potential applications for such evaporation energy-harvesting devices include
environmental sensors powered by the
natural environment. In
theory, such spore devices can generate more power per unit area than a
wind farm.[3] This research was funded by the
U.S. Department of Energy.[3]
References:
- Data Page for Isopropyl Alcohol, NIST.
- Xi Chen, Davis Goodnight, Zhenghan Gao, Ahmet H. Cavusoglu, Nina Sabharwal, Michael DeLay, Adam Driks, and Ozgur Sahin, "Scaling up nanoscale water-driven energy conversion into evaporation-driven engines and generators," Nature Communications, vol. 6, article no. 7346 (June 16, 2015), doi:10.1038/ncomms8346. This is an open access publication with a PDF file available here.
- Renewable energy from evaporating water, Columbia University Press Release, June 16, 2015.
- Renewable Energy from Evaporating Water, YouTube Video by ExtremeBio, June 16, 2015.
- Xi Chen, L. Mahadevan, Adam Driks, and Ozgur Sahin, "Bacillus spores as building blocks for stimuli-responsive materials and nanogenerators," Nature Nanotechnology, vol. 9, no. 2 (2014), http://dx.doi.org/10.1038/nnano.2013.290.
- Beth Kwon, "Biophysicist Harnesses Power of Evaporation, Discovers Potential New Source of Renewable Energy," Columbia University Press Release, January 28, 2014.
- Bacterial Spores Harness Evaporation Energy, YouTube Video by extremebio.org, January 27, 2014.
- Extremebio.org Web Site.
Permanent Link to this article
Linked Keywords: Thermodynamics; evaporation; high school; chemistry; teacher; salary; family; rent; apartment; duplex; two-family house; house; hydronic boiler; heating; thermostat; temperature; trick; alcohol; physics; physical mechanism; latent heat; vaporization; enthalpy of vaporization; heat capacity; Honeywell; patent; US Patent No. 2,394,920; sealed mercury switch; bimetallic strip; Wikimedia Commons; isopropyl alcohol; standard conditions for temperature and pressure; STP; kelvin; K; atmosphere; atm; kilojoule per mole; rubbing alcohol; mixture; water; milliliter; mole; calculation; concentration; volume; aqueous solution; approximation; estimate; calculation; Celsius; °C; Fahrenheit; energy conversion efficiency; inefficient; energy; thermopile; electric current; bar; tropical cyclone; hurricane; thermoelectric generator; radio; vodka; enthalpy of vaporization; vapor pressure; room temperature; drinking bird; felt; beak; dichloromethane; condensation; condense; scientist; engineer; Columbia University; microbiologist; Loyola University Chicago; mechanical energy; endospore; bacterial spore; humidity; force; summer; rainfall; nature; natural; phenomenon; energy transfer; Earth's climate; Ozgur Sahin; associate professor; biological sciences; author; wind; wave; ocean; research; material; chemistry; chemical; biomimetics; biomimetic system; oscillation; oscillate; fluid; work; actuator; Harvard University; Bacillus; gradient; human muscle; chemical synthesis; synthetic; bacterial spore; Bacillus bacteria; soil; microorganism; YouTube Video; self-preservation; survival mechanism; starvation; starved; reversible process; wood; plastic; mutation; self-assembly; self-assemble; submicrometer; substrate; elastomer; silicon; microcantilever; energy-harvesting; deflection; flexure; membrane; electricity; magnet; solenoid; coil of wire; Lego®; natural rubber; Xi Chen; scientific literature; publish; motion; locomotion; piston; rotation; rotary; electric power; light-emitting diode; gram; model car; miniature car; turbine; engine; fuel; paper; device architecture; artificial muscle; printing; parallel; shutter; environment; environmental; sensor; natural environment; scientific theory; wind farm; U.S. Department of Energy.